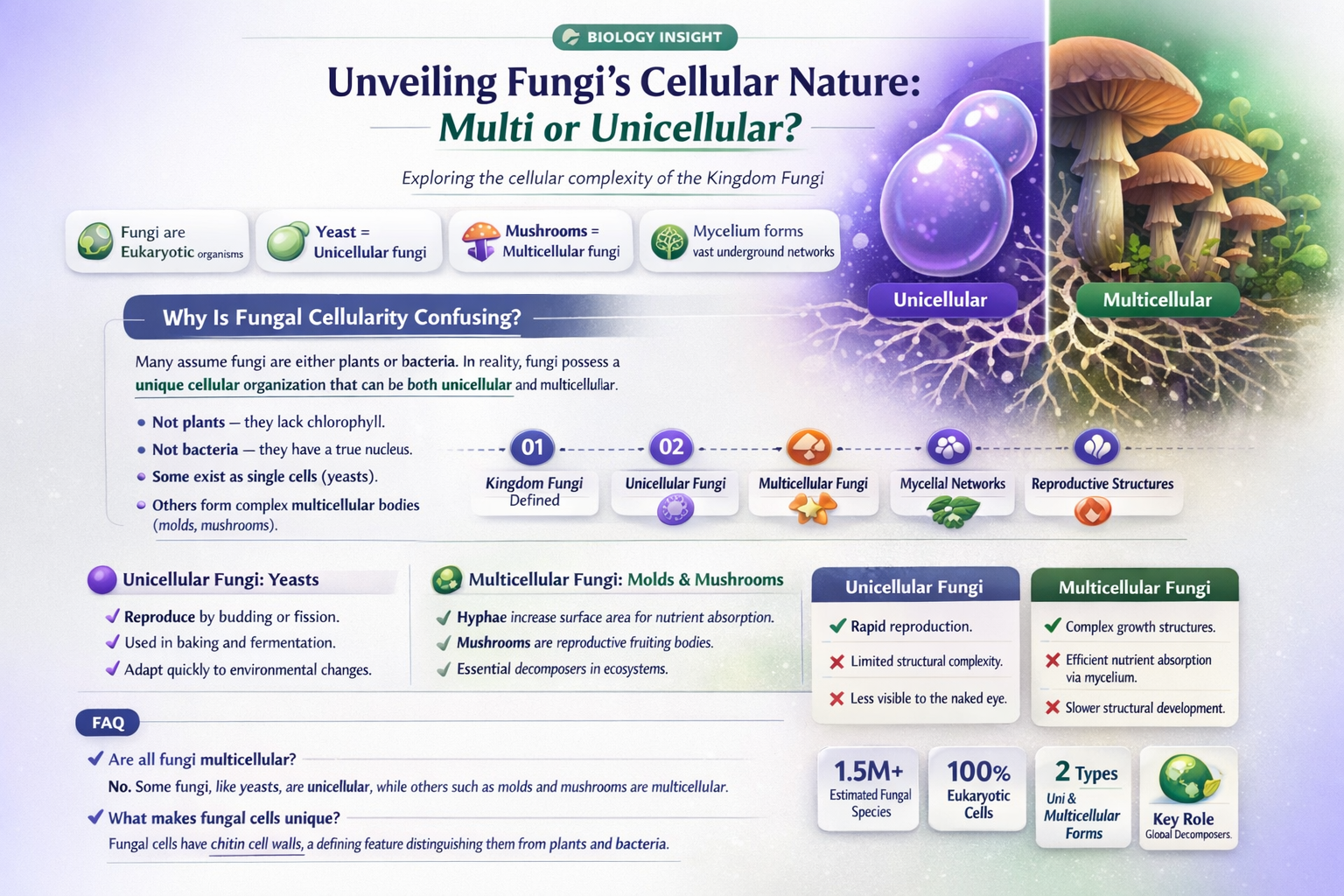
Fungi are a diverse group of organisms that play crucial roles in ecosystems, ranging from decomposers to symbionts and pathogens. One of the fundamental questions about fungi is whether they are multicellular or unicellular. While some fungi, like yeasts, exist as single-celled organisms, many others, such as mushrooms and molds, are multicellular, composed of a network of thread-like structures called hyphae. This duality highlights the complexity and adaptability of fungi, as they can thrive in various environments by adopting either unicellular or multicellular forms depending on their life cycle stage and ecological niche.
| Characteristics | Values |
|---|---|
| Cellular Nature | Both multi and unicellular |
| Unicellular Forms | Yeasts (e.g., Saccharomyces cerevisiae) |
| Multicellular Forms | Molds, mushrooms, and other filamentous fungi |
| Structural Units | Unicellular: single cell; Multicellular: hyphae (thread-like structures) or fruiting bodies |
| Reproduction | Unicellular: budding or fission; Multicellular: spores or fragmentation |
| Examples of Unicellular Fungi | Candida, Cryptococcus |
| Examples of Multicellular Fungi | Aspergillus, Penicillium, Agaricus (mushrooms) |
| Habitat | Diverse, including soil, water, and living organisms |
| Ecological Roles | Decomposers, symbionts, pathogens |
| Cell Walls | Present, primarily composed of chitin |
| Nutrition | Heterotrophic, absorbing nutrients from environment |
| Kingdom Classification | Fungi (separate from plants, animals, and bacteria) |
| Complexity | Ranges from simple unicellular to complex multicellular organisms |
Explore related products
$200.96 $278.25
What You'll Learn

Yeast as Unicellular Fungi
Yeast, a household name in baking and brewing, stands as a prime example of unicellular fungi. Unlike their multicellular counterparts, such as mushrooms or molds, yeast exists as individual cells, each capable of performing all life functions independently. This microscopic organism, belonging to the kingdom Fungi, thrives in diverse environments, from the sugary depths of fruit to the controlled conditions of laboratory cultures. Its unicellular nature grants yeast remarkable adaptability, enabling it to reproduce rapidly through budding or fission, a process that doubles its population in a matter of hours under optimal conditions.
Consider the role of yeast in fermentation, a metabolic process where sugars are converted into alcohol and carbon dioxide. In baking, a mere ¼ teaspoon of active dry yeast per cup of flour can leaven dough, creating the airy texture of bread. Brewers, on the other hand, often use a starter culture containing millions of yeast cells per milliliter to ferment sugars in wort, producing beer. This efficiency underscores yeast’s ability to function effectively as a single-celled organism, driving biochemical reactions without the need for cellular specialization seen in multicellular fungi.
From a practical standpoint, understanding yeast’s unicellular nature is crucial for its application in food and biotechnology. For home bakers, maintaining the correct temperature (ideally 75–80°F or 24–27°C) ensures yeast remains active, as extreme heat or cold can denature its proteins. In industrial settings, yeast strains like *Saccharomyces cerevisiae* are genetically engineered to produce insulin, vaccines, and biofuels, showcasing the versatility of this single-celled fungus. Its simplicity, combined with metabolic prowess, makes yeast an indispensable tool in both traditional and cutting-edge fields.
Comparatively, while multicellular fungi rely on complex networks of hyphae to absorb nutrients, yeast’s unicellular structure allows it to directly uptake resources from its environment. This efficiency is particularly evident in its ability to survive in nutrient-poor conditions, such as on the surface of fruits or in soil. Unlike multicellular fungi, which often form visible colonies like mold patches, yeast colonies appear as smooth, creamy masses, reflecting their uniform cellular structure. This distinction highlights the evolutionary advantage of unicellularity in certain ecological niches.
In conclusion, yeast exemplifies the functional elegance of unicellular fungi. Its ability to ferment, reproduce rapidly, and adapt to varied environments makes it a cornerstone of both natural ecosystems and human industries. Whether in a kitchen or a lab, yeast’s unicellular nature is not a limitation but a strength, offering simplicity, efficiency, and boundless potential. By harnessing this microscopic powerhouse, we unlock possibilities that span from artisanal bread to life-saving medicines.
Unveiling the Fungal Mystery: Identifying Milady's Unique Fungus Type
You may want to see also
Explore related products

Mold: Multicellular Fungal Structures
Fungi exhibit a remarkable diversity in their cellular organization, ranging from unicellular yeasts to complex multicellular mushrooms. Among these, molds stand out as quintessential examples of multicellular fungal structures. Unlike their unicellular counterparts, molds develop intricate networks of filamentous cells called hyphae, which collectively form a mycelium. This multicellular arrangement allows molds to efficiently colonize substrates, extract nutrients, and reproduce, making them highly successful in diverse environments.
Consider the lifecycle of *Aspergillus*, a common mold genus. It begins with a single spore germinating into a hypha, which then branches extensively to form a mycelium. This network is not merely a collection of individual cells but a coordinated system where nutrients and signals are shared. For instance, when part of the mycelium encounters a nutrient-rich area, it can redistribute resources to support growth in less favorable regions. This cooperative behavior is a direct consequence of multicellularity and highlights the adaptive advantages of mold structures.
From a practical standpoint, understanding mold’s multicellular nature is crucial for effective control and prevention. For example, surface cleaning may remove visible mold, but without addressing the underlying mycelium, regrowth is inevitable. To eradicate mold, one must target the entire structure, often requiring professional remediation techniques such as HEPA vacuuming and antimicrobial treatments. Homeowners should also monitor humidity levels (ideally below 60%) and promptly fix leaks, as molds thrive in damp conditions. These measures disrupt the multicellular network, preventing further colonization.
Comparatively, molds’ multicellular strategy contrasts with unicellular fungi like *Saccharomyces cerevisiae* (baker’s yeast), which rely on individual cells for survival and reproduction. While yeasts can quickly adapt to environmental changes through rapid cell division, molds invest in long-term persistence through their extensive hyphal networks. This difference explains why molds are often more challenging to eliminate in indoor environments compared to yeast contaminants. By recognizing this distinction, one can tailor interventions to the specific fungal structure at hand.
In conclusion, molds exemplify the sophistication of multicellular fungal structures through their hyphal networks and cooperative behaviors. Their ability to efficiently colonize and persist underscores the importance of targeted strategies for control. Whether in scientific research or household management, appreciating the unique characteristics of mold’s multicellularity is key to addressing fungal challenges effectively.
Yeast vs. Mold: Understanding the Key Differences and Uses
You may want to see also
Explore related products

Mushrooms: Complex Multicellular Examples
Mushrooms, often the poster children of the fungal kingdom, are a testament to the complexity of multicellular life in fungi. Unlike their unicellular cousins, such as yeast, mushrooms develop intricate structures that include a network of thread-like hyphae, a stalk (stipe), and a cap (pileus) with gills or pores for spore production. This level of organization allows them to efficiently decompose organic matter, recycle nutrients, and form symbiotic relationships with plants. For instance, mycorrhizal mushrooms partner with tree roots to enhance nutrient uptake, showcasing their ecological significance. Understanding their multicellular nature is key to appreciating their role in ecosystems and their potential applications in medicine, agriculture, and biotechnology.
To cultivate mushrooms at home, start by selecting a species suited to your environment—oyster mushrooms thrive in cooler temperatures, while shiitake prefer warmer conditions. Purchase spore-infused substrate (mycelium-colonized material) or grow kits for beginners. Maintain humidity levels around 80-90% by misting the growing area daily, and ensure proper ventilation to prevent mold. Harvest mushrooms when the caps are fully open but before spores drop, typically 5-7 days after fruiting begins. This hands-on approach not only yields fresh produce but also highlights the coordinated growth of their multicellular structures.
From a comparative perspective, mushrooms stand apart from other multicellular organisms due to their chitinous cell walls and heterotrophic lifestyle. Unlike plants, they lack chlorophyll and obtain nutrients by breaking down organic matter externally. Their hyphae form a mycelium network, often referred to as the "wood wide web," which can span acres underground. This network facilitates communication and resource sharing among plants and fungi, demonstrating a level of complexity akin to neural networks. Such adaptations underscore why mushrooms are not just simple organisms but sophisticated multicellular entities.
Persuasively, mushrooms’ multicellular nature makes them invaluable in addressing global challenges. Their mycelium can degrade pollutants like oil and plastics, offering eco-friendly remediation solutions. In medicine, compounds like psilocybin from *Psilocybe* species are being researched for mental health treatments, with clinical trials showing promise in alleviating depression and anxiety. Even in construction, mycelium-based materials are emerging as sustainable alternatives to foam and leather. By harnessing their complexity, we can unlock innovations that benefit both humanity and the planet.
Descriptively, the lifecycle of a mushroom reveals its multicellular mastery. Beginning as a spore, it germinates into hyphae, which intertwine to form mycelium. Under optimal conditions, the mycelium aggregates to create a primordium, the precursor to the mushroom’s fruiting body. As the stipe elongates and the cap expands, gills or pores develop to release spores, completing the cycle. This orchestrated process, driven by cellular differentiation and communication, exemplifies the sophistication of multicellular fungi. Observing this lifecycle firsthand, whether in a forest or a grow kit, offers a profound appreciation for the intricate biology of mushrooms.
Fungi's Role: Producer, Consumer, or Decomposer? Unraveling the Mystery
You may want to see also
Explore related products

Dimorphic Fungi: Switching Cell Types
Fungi exhibit a remarkable diversity in their cellular organization, existing as both unicellular and multicellular organisms. Among this kingdom, a subset known as dimorphic fungi stands out for their ability to switch between two distinct cell types—yeast and mold—in response to environmental cues. This adaptability is not merely a biological curiosity but a critical survival mechanism that influences their pathogenicity, ecology, and interactions with hosts. Understanding this phenomenon is essential for fields ranging from medicine to agriculture, as it sheds light on how these organisms thrive in varying conditions.
Consider the fungus *Histoplasma capsulatum*, a prime example of dimorphism in action. In the environment, it grows as a filamentous mold, producing spores that can become airborne. Upon inhalation by a human host, the warmer body temperature (37°C or 98.6°F) triggers a transformation into yeast cells, which are better suited for intracellular survival and evasion of the immune system. This switch is not just morphological but involves significant genetic and metabolic changes, such as altered cell wall composition and enzyme production. For instance, the cell wall thickens in yeast form, providing resistance to host defenses. Clinically, this behavior complicates treatment, as antifungal agents must target both forms effectively.
The mechanism behind this transition is tightly regulated by temperature, oxygen levels, and nutrient availability. For example, *Coccidioides immitis*, another dimorphic fungus, requires a temperature shift from 25°C to 37°C to convert from mold to yeast. Researchers have identified specific genes, such as those encoding heat-shock proteins and transcription factors, that play pivotal roles in this process. Manipulating these genes could offer novel therapeutic strategies, such as disrupting the yeast phase to reduce pathogenicity. However, such interventions must be precise, as off-target effects could harm beneficial fungi or even human cells.
From a practical standpoint, recognizing dimorphism is crucial for accurate diagnosis and treatment of fungal infections. For instance, histoplasmosis, caused by *H. capsulatum*, may present as a mild respiratory illness or severe systemic disease, depending on the host’s immune status. Laboratory identification often involves culturing the fungus at different temperatures to observe the phase transition. Patients at higher risk, such as immunocompromised individuals or those in endemic areas, should undergo early screening, which may include serum antigen tests or chest imaging. Prophylactic measures, like avoiding dusty environments where fungal spores are prevalent, can reduce exposure risk.
In conclusion, dimorphic fungi exemplify the dynamic nature of fungal biology, showcasing how environmental cues dictate cellular identity. Their ability to switch between unicellular and multicellular forms is a testament to evolutionary ingenuity, with profound implications for health and disease. By studying these transitions, scientists can develop targeted therapies and preventive strategies, ensuring that these adaptable organisms are managed effectively in both clinical and environmental contexts. Whether in the lab or the field, understanding dimorphism is key to unlocking the complexities of the fungal kingdom.
Is Coral a Fungus? Unraveling the Mystery of Coral's True Nature
You may want to see also

Role of Hyphae in Multicellularity
Fungi exhibit a remarkable diversity in their cellular organization, ranging from unicellular yeasts to complex multicellular mushrooms. At the heart of fungal multicellularity lies the hypha, a filamentous structure that serves as the fundamental building block of many fungal species. Hyphae are long, thread-like cells that grow by apical extension, allowing fungi to explore and colonize their environment efficiently. But what makes hyphae so crucial to the multicellular nature of fungi?
Consider the structural and functional advantages hyphae provide. Unlike unicellular organisms, which rely on individual cells to perform all life functions, hyphae enable a division of labor within the fungal organism. For instance, some hyphae specialize in nutrient absorption, while others focus on structural support or reproduction. This modularity is akin to the organ systems in higher organisms, showcasing a sophisticated level of organization. In practical terms, this means a fungus can simultaneously degrade organic matter, transport nutrients, and produce spores, all through the coordinated activity of its hyphal network.
To understand the role of hyphae in multicellularity, imagine a fungal colony as a city. Each hypha functions like a road or utility line, connecting different parts of the colony and facilitating the exchange of resources. This interconnectedness allows fungi to thrive in diverse environments, from soil to decaying wood. For example, mycorrhizal fungi use their hyphal networks to form symbiotic relationships with plant roots, enhancing nutrient uptake for both partners. Without hyphae, such complex interactions would be impossible, underscoring their role as the backbone of fungal multicellularity.
However, the benefits of hyphae come with challenges. Maintaining a multicellular structure requires coordination and communication between cells. Fungi achieve this through chemical signaling and the transport of molecules via the hyphal network. For instance, when a hypha detects a nutrient source, it can relay this information to other parts of the colony, directing growth toward the resource. This level of integration is a hallmark of multicellularity and highlights the adaptive advantages hyphae confer.
In conclusion, hyphae are not merely structural components but dynamic systems that enable fungi to achieve multicellularity. Their ability to grow, differentiate, and communicate makes them indispensable to the fungal lifestyle. By studying hyphae, we gain insights into the evolutionary strategies that allow fungi to dominate ecosystems as decomposers, pathogens, and symbionts. Whether you’re a researcher, educator, or simply curious about the natural world, understanding the role of hyphae offers a deeper appreciation for the complexity of fungal life.
Shared Secrets: Uniting Humans, Plants, and Mushrooms in Nature's Web
You may want to see also
Frequently asked questions
Fungi can be both multi and unicellular. Some fungi, like yeasts, are unicellular, while others, such as mushrooms and molds, are multicellular.
Examples of unicellular fungi include yeasts, such as *Saccharomyces cerevisiae*, which is commonly used in baking and brewing.
Examples of multicellular fungi include mushrooms, molds (e.g., *Penicillium*), and bracket fungi, which form complex structures like mycelium and fruiting bodies.
Unicellular fungi consist of a single cell, while multicellular fungi are composed of many cells organized into structures like hyphae (thread-like filaments) and mycelium (a network of hyphae).

































